|
11/5/2022 0 Comments Formula for percent by mmass  Make sure that the mass units match before solving. We can substitute the mass of the active ingredient and the mass of the anti-inflammatory ointment into the key equation. Mass Percent (Mass of Solute / Mass of Solution)100 Mass (m solute / mSolution)100 This formula uses 3 Variables Variables Used Mass Percent - Mass Percent is a way of expressing a concentration or describing the component in a particular mixture. In this problem, the main active ingredient is the solute, and the anti-inflammatory ointment is the solution. The solvent is the substance doing the dissolving, and the solution is a mixture produced by combining the solute and solvent. The solute is the substance that is dissolved. To use this equation, we should recognize a few key terms. Step-2: Divide the increased value by the original number and further multiply the answer by 100. You’ll determine the mass percentage of every element with these masses. Get the mass of each element by assuming a certain overall mass for the sample (100 g is a good mass to assume when working with percentages). Thus to calculate the percentage increase we will follow two steps: Step-1: Calculate the difference i.e. Percentage Concentration of Solute in any Solution (percentage weight by volume formula) The mass of solute in per ml volume of solution 100. In order to find the percent by mass of the active ingredient in this ointment, we must recognize the key equation: percent concentration by mass is equal to the mass of the solute divided by the mass of the solution times 100 percent. Mass percent (component’s mass total mass) x 100 or Percentage of mass (solute’s mass mass of solution) x 100 The Mass percent formula is expressed as solving for the molar mass also for the mass of every element in 1 mole of the compound. What is the percent by mass of the active ingredient in this ointment? If you are given mass percentages, always work with a hypothetical 100-gram sample. Enter a valid molecular formula and press the calculate button to determine. To make the calculation easy (i.e., let the percentages convert directly to grams), let's assume we have 100 g of vitamin C. Determine the Molar mass and Percent mass composition of a linear chemical formula.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

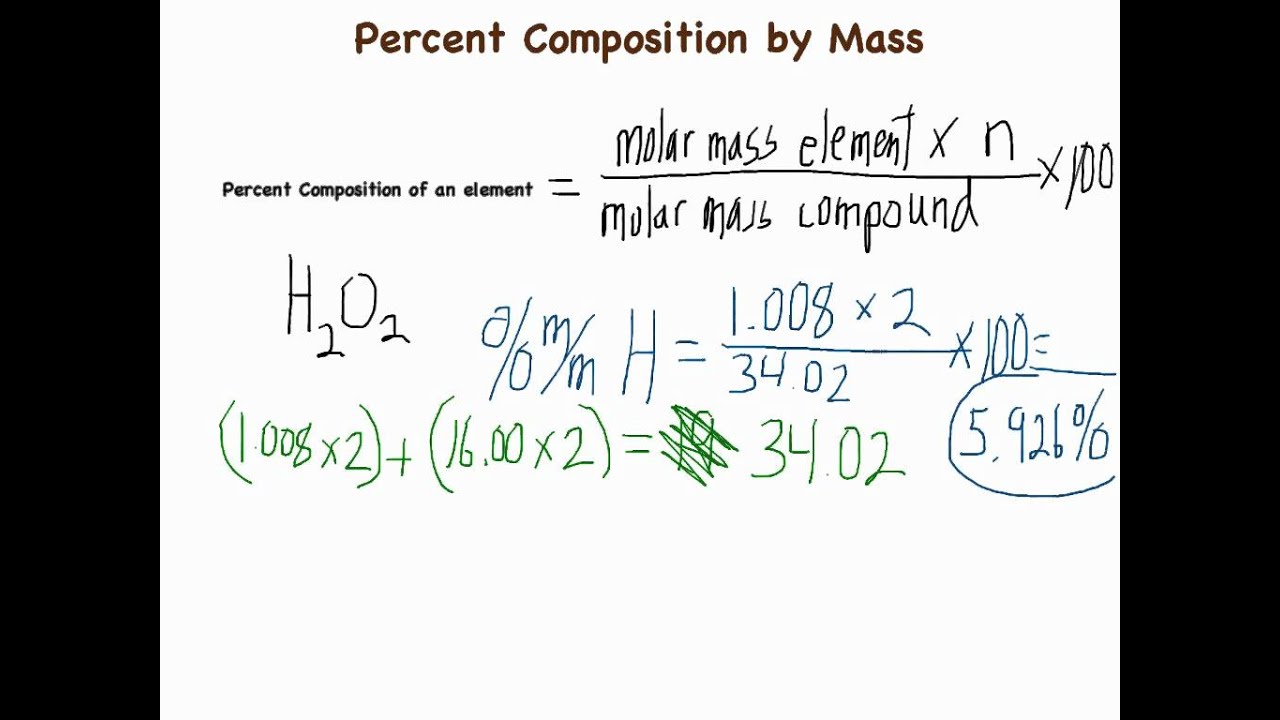
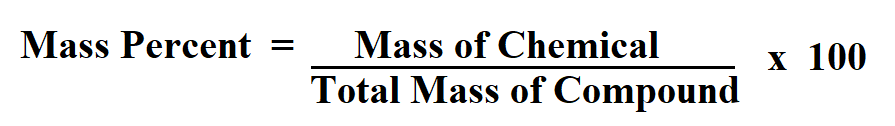
 RSS Feed
RSS Feed
